Oxygen charge 16/23/2023  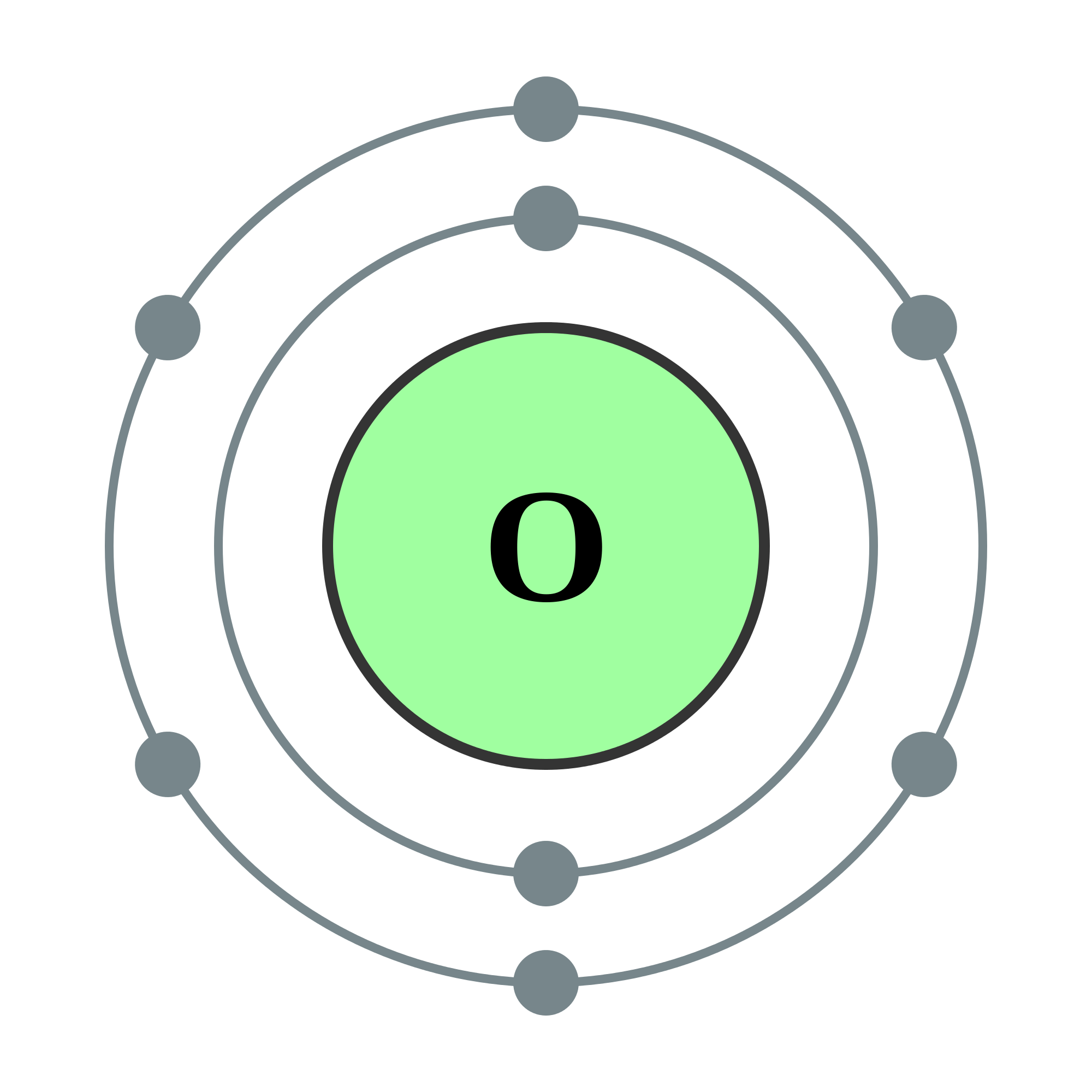
Consider the Lewis structure of methanol, CH 3OH (methanol is the so-called ‘wood alcohol’ that unscrupulous bootleggers sometimes sold during the prohibition days in the 1920's, often causing the people who drank it to go blind). Recognizing and distinguishing between neutral and charged bonding patterns will be helpful in learning reaction mechanisms. For example, for NO3, the formal charge on the nitrogen and oxygen atoms must add up to 1. During chemical reactions, it is common to have charge reactant, intermediates, and/or products. in the direction perpendicular to the laser beam using an intensified charge. Organic molecules can also have positive or negative charges associated with them. The plasma is ignited in a gas channel of 1 × 1 mm2 cross section and 30 mm. In the beginning, it can be helpful to physically add the lone pair electrons.įor organic chemistry, the common bonding patterns of carbon, oxygen, and nitrogen have useful applications when evaluating chemical structures and reactivity. Since the lone pair electrons are often NOT shown in chemical structures, it is important to mentally add the lone pairs. Unshared electrons are also called ‘Lone Pairs’ and are shown as ‘:’ NH2OH is made up of one nitrogen (N), three hydrogens (H), and one oxygen. 1 2 In simple terms, formal charge is the difference. Double and triple bonds can also be communicated with lines as shown below.Ģ shared electrons form a single bond shown as ‘:’ or ‘–‘Ĥ shared electrons form a double bond shown as ‘::’ or ‘=’Ħ shared electrons form at triple bond shown as ‘:::’ or Hence formal charge on I atom in IF2-7-4/2-6 -1 Formal charge on each F atom. or q ), in the covalent view of chemical bonding, is the hypothetical charge assigned to an atom in a molecule, assuming that electrons in all chemical bonds are shared equally between atoms, regardless of relative electronegativity. Lone pair (unshared) electrons are still shown as individual electrons. 
Line representations are only used for shared electrons. While it can be helpful initially to write the individual shared electrons, this approach quickly becomes awkward.Ī single line is used to represent one pair of shared electrons. Lewis structures, also known as Lewis-dot diagrams, show the bonding relationship between atoms of a molecule and the lone pairs of electrons in a molecule. Often, an oxygen atom will have only 1 bond. Note: The review of general chemistry in sections 1.3 - 1.6 is integrated into the above Learning Objective for organic chemistry in sections 1.7 and 1.8. Oxygen will usually have 2 bonds, occasionally 3 however, if the O has 3 bonds it will be positively charged. \)ĭraw, interpret, and convert between Lewis (Kekule), Condensed, and Bond-line Structures Amazfit GTR 4 Smart Watch for Men Android iPhone, Dual-Band GPS Alexa, Bluetooth Call, 150 Sports Modes, 14-Day Battery Life, Heart Rate Blood Oxygen Monitor, 1.43AMOLED Display,Black (Renewed) Amazfit GTS 4 Smart Watch for Men, Dual-Band GPS, Alexa Built-in, Bluetooth Calls, 150 Sports Modes, Heart Rate SPO Monitor, 1.75 AMOLED. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
